 For Laboratory Use Only: Intended strictly for research & development, and quality control purposes by qualified personnel only. Not for human or animal consumption, or for any medical, therapeutic, or diagnostic use.
For Laboratory Use Only: Intended strictly for research & development, and quality control purposes by qualified personnel only. Not for human or animal consumption, or for any medical, therapeutic, or diagnostic use.
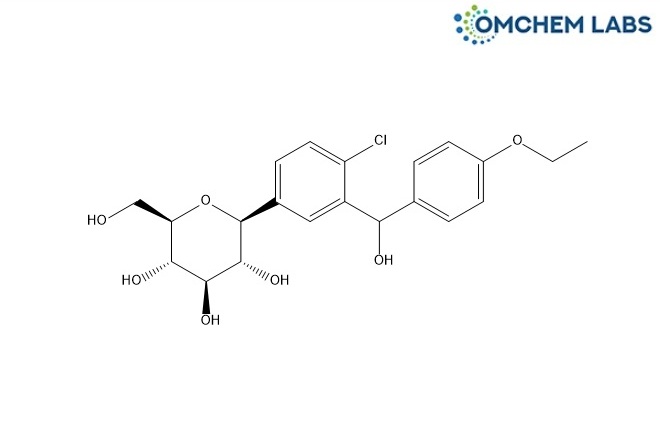
Dapagliflozin Hydroxy Benzylic Impurity
| Catalogue No |
DAPA-OCL-001 |
| CAS NO |
1204222-85-4 |
| Molecular Formula | C21H25ClO7 |
| Molecular weight | 424.87 |
| Inquiry Status | In Stock |
| Synonyms | (2S,3R,4R,5S,6R)-2-(4-Chloro-3-((4-ethoxyphenyl)(hydroxy)methyl)phenyl)-6-(hydroxymethyl)tetrahydro-2H-pyran-3,4,5-triol |
Detailed Overview of this Impurity: Discover more about Impurity Standard & Analysis
Impurity Profiling of Dapagliflozin Hydroxy Benzylic Impurity
Introduction
Impurity profiling is a central component of modern pharmaceutical quality assessment, ensuring that drug substances remain safe, stable, and compliant with regulatory expectations. The study of Dapagliflozin Hydroxy Benzylic Impurity represents an important example of how structurally related impurities can emerge during the lifecycle of an active pharmaceutical ingredient. Such impurities may influence physicochemical behavior, stability, or pharmacological performance, making their identification and control an essential aspect of drug development. A structured evaluation combining process understanding, analytical investigation, and purification control forms the basis of reliable impurity management.
Formation of Impurities During API Synthesis
The generation of Dapagliflozin Hydroxy Benzylic Impurity can typically be associated with transformation events occurring along the synthetic pathway of the parent compound. Variations in reaction selectivity, intermediate stability, or reagent compatibility may encourage the formation of structurally related species. Oxidative processes, rearrangements, or incomplete conversions may also contribute to impurity development. In addition, environmental influences such as moisture exposure, light interaction, or thermal stress during processing and storage can further modify molecular structures, producing secondary impurities that must be monitored carefully. A detailed understanding of reaction mechanisms therefore supports proactive impurity control.
Analytical Data Interpretation Techniques
Accurate identification of Dapagliflozin Hydroxy Benzylic Impurity depends on the integration of multiple analytical techniques. Chromatographic approaches allow separation of closely related molecular entities, while spectroscopic tools provide structural insights based on molecular signatures. Interpretation of chromatographic profiles, spectral patterns, and fragmentation behavior enables scientists to distinguish the impurity from the primary compound and other related species. This combined analytical perspective supports consistent impurity tracking across production batches and enhances process transparency.
Method Validation for Impurity Detection
Reliable impurity assessment requires that analytical procedures used for detecting Dapagliflozin Hydroxy Benzylic Impurity undergo systematic validation. Validation confirms that a method is capable of selectively identifying the impurity while maintaining accuracy and reproducibility under routine conditions. Key performance attributes such as specificity, consistency, and robustness are evaluated to ensure that the method remains dependable across different analysts, instruments, and production stages. Such validation establishes confidence in impurity monitoring and supports regulatory documentation.
Purification Strategies for Reducing Impurities
Once identified, impurity levels must be effectively controlled through suitable purification strategies. For compounds like Dapagliflozin Hydroxy Benzylic Impurity, purification approaches often rely on differences in solubility, polarity, or molecular interaction behavior. Techniques including selective crystallization, solvent-based refinement, and chromatographic separation can be optimized to enhance product purity. Process refinement at this stage not only reduces impurity carryover but also improves reproducibility and scalability of manufacturing operations.
Isolation and Characterization of Impurities
When structural confirmation is required, Dapagliflozin Hydroxy Benzylic Impurity may be isolated in a controlled manner to allow comprehensive characterization. Isolation facilitates direct examination of molecular structure using advanced spectroscopic tools, enabling confirmation of chemical identity and assessment of potential impact. This step is essential for establishing reference standards, understanding degradation pathways, and supporting toxicological evaluation where necessary. Proper characterization strengthens overall impurity knowledge and improves lifecycle control strategies.
Conclusion
The profiling of Dapagliflozin Hydroxy Benzylic Impurity highlights the importance of integrating synthetic insight, analytical capability, validation rigor, and purification efficiency into a unified quality framework. Through systematic monitoring and structural understanding, impurities can be effectively managed to maintain the consistency and safety of pharmaceutical products. A proactive impurity strategy ultimately contributes to regulatory confidence, manufacturing reliability, and patient protection throughout the product lifecycle.
