 For Laboratory Use Only: Intended strictly for research & development, and quality control purposes by qualified personnel only. Not for human or animal consumption, or for any medical, therapeutic, or diagnostic use.
For Laboratory Use Only: Intended strictly for research & development, and quality control purposes by qualified personnel only. Not for human or animal consumption, or for any medical, therapeutic, or diagnostic use.
Dapagliflozin is an oral antidiabetic medication belonging to the sodium-glucose co-transporter-2 (SGLT2) inhibitor class, commonly used in the management of type 2 diabetes mellitus, heart failure, and chronic kidney disease by promoting renal glucose excretion and improving glycemic control. In addition to its metabolic benefits, it has demonstrated cardiovascular and renal protective effects in clinical practice. Maintaining the pharmaceutical purity of Dapagliflozin is essential to ensure patient safety, therapeutic effectiveness, and regulatory compliance. Impurity profiling plays a key role in detecting and controlling process-related impurities, degradation products, and residual solvents that may arise during synthesis, formulation, or storage. Here are some of its known impurities listed below.
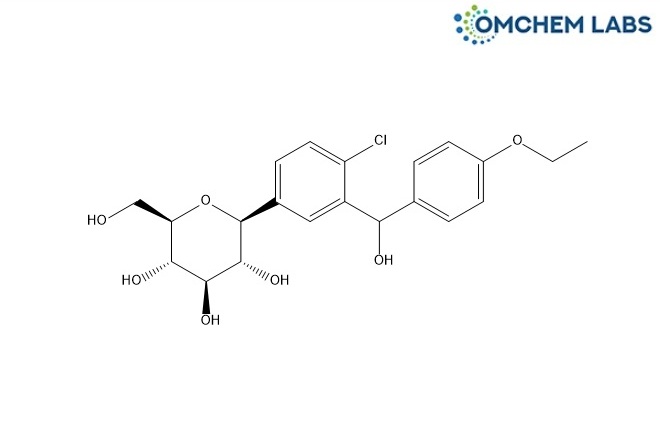
CAS No : 1204222-85-4
(2S,3R,4R,5S,6R)-2-(4-Chloro-3-((4-ethoxyphenyl)(hydroxy)methyl)phenyl)-6-(hydroxymethyl)tetrahydro-2H-pyran-3,4,5-triol
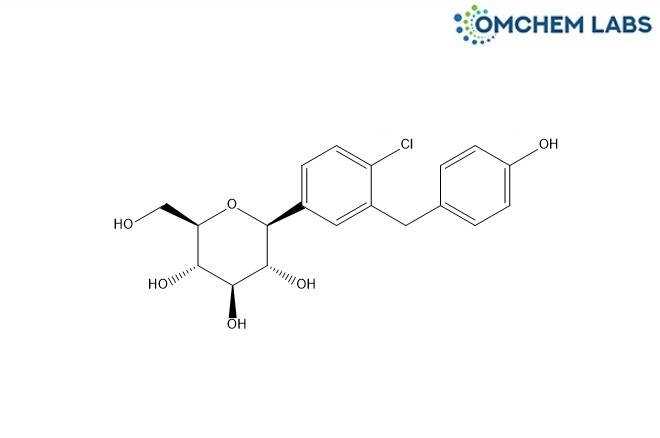
CAS No : 864070-37-1
(2S,3R,4R,5S,6R)-2-(4-Chloro-3-(4-hydroxybenzyl)phenyl)-6-(hydroxymethyl)tetrahydro-2H-pyran-3,4,5-triol
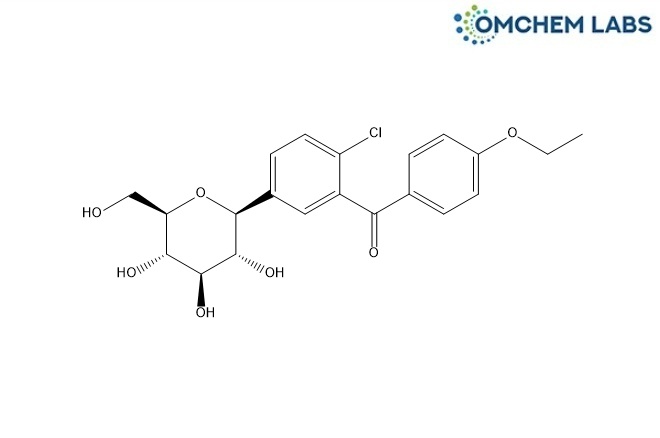
CAS No : 2169998-23-4
(2-Chloro-5-((2S,3R,4R,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)phenyl)(4-ethoxyphenyl)methanone
