 For Laboratory Use Only: Intended strictly for research & development, and quality control purposes by qualified personnel only. Not for human or animal consumption, or for any medical, therapeutic, or diagnostic use.
For Laboratory Use Only: Intended strictly for research & development, and quality control purposes by qualified personnel only. Not for human or animal consumption, or for any medical, therapeutic, or diagnostic use.
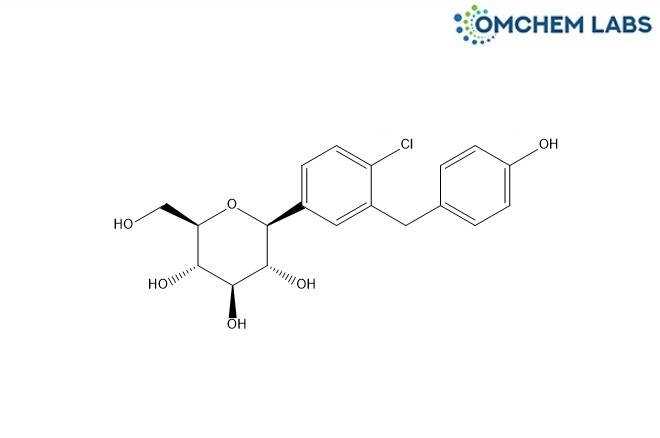
Desethyl Dapagliflozin
| Catalogue No |
DAPA-OCL-002 |
| CAS NO |
864070-37-1 |
| Molecular Formula | C19H21ClO6 |
| Molecular weight | 380.82 |
| Inquiry Status | In Stock |
| Synonyms | (2S,3R,4R,5S,6R)-2-(4-Chloro-3-(4-hydroxybenzyl)phenyl)-6-(hydroxymethyl)tetrahydro-2H-pyran-3,4,5-triol |
Detailed Overview of this Impurity: Discover more about Impurity Standard & Analysis
Impurity Profiling of Desethyl Dapagliflozin Impurity: A Scientific Overview
Introduction
Impurity profiling plays a central role in modern pharmaceutical quality systems, where even trace-level components must be understood and controlled. The study of Desethyl Dapagliflozin Impurity is relevant not only for regulatory compliance but also for ensuring consistent therapeutic performance of the associated active pharmaceutical ingredient. Impurities may arise from synthetic transformations, raw material variability, or environmental exposure, and their presence can influence stability, safety, and manufacturability. A systematic evaluation of their origin, detection, and control therefore forms an integral part of API lifecycle management.
Formation of Impurities During API Synthesis
During multistep synthesis routes, compounds structurally related to the target molecule may form through incomplete reactions, secondary pathways, or reagent interactions. In the context of Desethyl Dapagliflozin Impurity, such species may originate from transformation of intermediates, rearrangements occurring under specific reaction environments, or modification of functional groups during process optimization. Variations in solvent composition, catalytic conditions, or reaction time can subtly influence these pathways. Additionally, downstream handling, exposure to moisture, or oxidative conditions may generate related compounds during storage or processing. Understanding these formation mechanisms supports better process control and predictive impurity management.
Analytical Data Interpretation Techniques
Accurate impurity profiling relies on the integration of complementary analytical platforms capable of resolving structurally similar species. Chromatographic separation methods provide insight into retention behavior and relative abundance, while spectroscopic tools assist in structural differentiation. For Desethyl Dapagliflozin Impurity, interpretation of chromatographic patterns, spectral features, and fragmentation trends allows analysts to distinguish process-related species from degradation products. Correlating these observations with synthetic knowledge helps establish impurity pathways and supports ongoing process monitoring. Consistent analytical interpretation also contributes to long-term manufacturing reproducibility.
Method Validation for Impurity Detection
To ensure that impurity data are reliable and defensible, analytical procedures must demonstrate fitness for purpose through formal validation. In the case of Desethyl Dapagliflozin Impurity, validated methods confirm that the analytical approach consistently detects, separates, and estimates related components under defined conditions. Validation activities typically assess method selectivity, reproducibility, sensitivity, and operational stability. Demonstrating robustness across small variations in analytical conditions ensures that impurity monitoring remains dependable during routine quality control and stability studies. Such validation practices reinforce confidence in the generated impurity profile.
Purification Strategies for Reducing Impurities
The reduction of undesired species often requires a combination of chemical and physical purification approaches tailored to molecular properties. For compounds related to Desethyl Dapagliflozin Impurity, purification may involve exploiting differences in solubility, polarity, or crystallization behavior between the impurity and the desired product. Controlled recrystallization, solvent adjustment, or chromatographic refinement may all contribute to improved purity. The optimization of these strategies must balance efficiency, yield, and scalability while preserving the integrity of the active compound. A well-designed purification workflow can significantly reduce impurity burden without introducing new variability.
Isolation and Characterization of Impurities
When detailed understanding is required, impurities are often isolated in enriched form to permit structural investigation. For Desethyl Dapagliflozin Impurity, preparative separation techniques enable collection of sufficient material for spectroscopic evaluation. Structural elucidation using complementary methods provides insight into functional group arrangement, molecular connectivity, and potential formation pathways. Such characterization supports toxicological assessment, informs process refinement, and aids in the development of reference standards for routine analysis. Comprehensive characterization ensures that impurity knowledge evolves alongside process development.
Conclusion
The profiling of Desethyl Dapagliflozin Impurity illustrates the broader scientific approach required for impurity management in pharmaceutical manufacturing. By integrating mechanistic understanding of synthesis, advanced analytical interpretation, validated detection methods, targeted purification strategies, and thorough structural characterization, manufacturers can maintain consistent product quality while meeting regulatory expectations. A proactive impurity control strategy not only strengthens compliance but also contributes to the overall reliability and safety of pharmaceutical therapies.
