 For Laboratory Use Only: Intended strictly for research & development, and quality control purposes by qualified personnel only. Not for human or animal consumption, or for any medical, therapeutic, or diagnostic use.
For Laboratory Use Only: Intended strictly for research & development, and quality control purposes by qualified personnel only. Not for human or animal consumption, or for any medical, therapeutic, or diagnostic use.
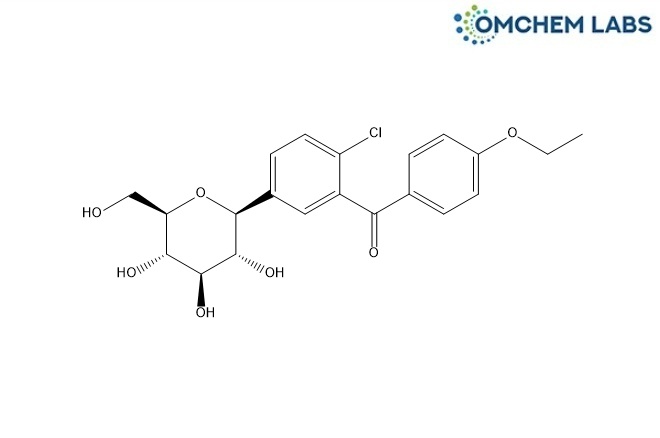
Oxo Dapagliflozin
| Catalogue No |
DAPA-OCL-003 |
| CAS NO |
2169998-23-4 |
| Molecular Formula | C21H23ClO7 |
| Molecular weight | 422.90 |
| Inquiry Status | In Stock |
| Synonyms | (2-Chloro-5-((2S,3R,4R,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)phenyl)(4-ethoxyphenyl)methanone |
Detailed Overview of this Impurity: Discover more about Impurity Standard & Analysis
Impurity Profiling of Oxo Dapagliflozin Impurity: A Scientific Overview of Formation, Detection, and Control
Introduction
The study of pharmaceutical impurities plays a pivotal role in ensuring the safety, performance, and regulatory acceptance of drug substances. The presence of structurally related compounds such as Oxo Dapagliflozin Impurity can arise from synthetic transformations, degradation pathways, or process variability. Understanding the origin, behavior, and detectability of such impurities is essential for maintaining consistent product quality. A well-designed impurity profiling strategy integrates synthetic insight, analytical evaluation, purification science, and structural characterization to support both development and lifecycle management of the parent compound.
Formation of Impurities During API Synthesis
Impurities related to compounds like Oxo Dapagliflozin Impurity may develop at multiple stages of a synthetic process. These can emerge from incomplete reactions, rearrangements, oxidation events, or competing side pathways. The selection of reagents, solvent systems, catalysts, and reaction conditions often determines the impurity landscape. In addition to process-driven origins, exposure to environmental factors during processing or storage may promote transformation of the parent molecule into secondary species. A thorough understanding of reaction mechanisms and material compatibility therefore becomes central to anticipating and controlling impurity formation.
Analytical Data Interpretation Techniques
Reliable detection of Oxo Dapagliflozin Impurity requires a combination of separation science and structural analysis. Chromatographic methods are typically employed to resolve closely related molecular species, while spectroscopic tools help reveal structural attributes. Interpretation of analytical data involves correlating retention behavior, fragmentation patterns, and spectral signatures with chemical structure. By integrating orthogonal techniques, analysts can distinguish the impurity from the principal compound and from other trace constituents, ensuring accurate representation of the impurity profile across production batches.
Method Validation for Impurity Detection
For impurity monitoring to be meaningful, analytical procedures must demonstrate consistent performance under defined conditions. Validation exercises confirm that the chosen method is capable of selectively detecting Oxo Dapagliflozin Impurity without interference from the matrix or related compounds. Parameters such as method selectivity, repeatability, sensitivity, and stability-indicating capability are typically evaluated. Establishing method robustness ensures that impurity monitoring remains reliable across different analysts, instruments, and production environments, which is essential for regulatory confidence.
Purification Strategies for Reducing Impurities
Controlling the presence of Oxo Dapagliflozin Impurity often involves refinement of downstream processing steps. Techniques based on differences in solubility, polarity, volatility, or molecular size can be leveraged to separate the impurity from the desired product. Processes such as selective crystallization, phase partitioning, or chromatographic purification may be optimized to enhance removal efficiency while maintaining yield. Process adjustments at this stage frequently contribute significantly to achieving the desired purity profile of the final API.
Isolation and Characterization of Impurities
When an impurity requires deeper understanding, isolation becomes necessary for structural confirmation and safety evaluation. Preparative-scale separation techniques allow collection of the impurity in sufficient purity for spectroscopic examination. Structural elucidation through complementary analytical methods provides insight into its chemical origin and potential reactivity. This information supports toxicological assessment, reference standard preparation, and the establishment of appropriate control strategies throughout manufacturing and storage.
Conclusion
Profiling impurities such as Oxo Dapagliflozin Impurity represents a multidisciplinary effort combining process chemistry, analytical science, purification engineering, and structural investigation. By systematically examining how impurities arise, how they are detected, and how they can be minimized, pharmaceutical developers can ensure consistent API quality and regulatory compliance. A proactive impurity management approach ultimately strengthens product reliability and contributes to the broader goal of safeguarding patient health.
